MiRNA Sequencing | Assay Market | Liquid Biopsy | Regional Breakdown | April 2026 | Source: Market Research
2.1B∗∗∣∗∗12.50.65B
Market Value by 2035 | CAGR (2026-2035) | Market Value in 2025
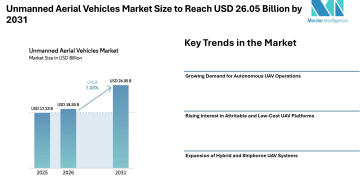
MiRNA Sequencing Assay Market
Key Takeaways
-
MiRNA Sequencing Assay Market is projected to reach USD 2.1 billion by 2035 at a 12.5% CAGR .
-
Next-generation sequencing (NGS) dominates the technology segment; real-time PCR (qPCR) is the fastest-growing .
-
Oncology and cancer research applications lead market demand; cardiovascular and neurological diseases are rapidly growing .
-
Illumina, Thermo Fisher Scientific, Qiagen, Agilent Technologies, and Bio-Rad lead the competitive landscape .
-
North America holds the largest market share; Asia-Pacific is the fastest-growing region .
The MiRNA Sequencing Assay Market is projected to grow from USD 0.65 billion in 2025 to USD 2.1 billion by 2035 at a 12.5% CAGR , driven by increasing prevalence of cancer and chronic diseases, growing demand for liquid biopsy-based diagnostics, and the adoption of MiRNA Sequencing and Liquid Biopsy technologies that enable non-invasive disease detection, early diagnosis, and personalized treatment monitoring across oncology, cardiovascular, and neurological applications worldwide .
Market Size and Forecast (2025-2035)
*Source: Market Research Future *
Segment & Technology Breakdown
*Source: Market Research Future *
What Is Driving the MiRNA Sequencing Assay Market Demand?
Increasing Cancer Prevalence: The global cancer burden continues to rise, with approximately 19.3 million new cases and 10 million cancer deaths reported in 2020. MiRNA sequencing offers non-invasive liquid biopsy alternatives to traditional tissue biopsies, driving demand for early detection and monitoring tools .
Advancements in Next-Generation Sequencing (NGS): NGS technologies have dramatically reduced sequencing costs from 100millionpergenomein2001tounder600 today. This cost reduction has made comprehensive miRNA profiling accessible to routine research and clinical applications .
Growing Liquid Biopsy Adoption: Liquid biopsy market is projected to reach $8 billion by 2028. MiRNA biomarkers in blood, urine, and other body fluids enable non-invasive cancer detection, treatment monitoring, and recurrence surveillance without surgical biopsies .
Rising Focus on Personalized Medicine: Healthcare systems are shifting from one-size-fits-all treatments to precision medicine approaches. MiRNA expression profiles predict drug response, disease progression, and patient outcomes, enabling tailored therapeutic strategies .
KEY INSIGHT
|> Next-Generation Sequencing (NGS) is the leading technology segment, offering high-throughput comprehensive profiling. Real-time PCR (qPCR) is the fastest-growing segment due to its cost-effectiveness and suitability for targeted validation studies. The consumables segment holds the largest market share, driven by recurring purchases of kits and reagents for miRNA isolation, library preparation, and detection .
Get the full data — free sample available:
→ Download Free Sample PDF: MiRNA Sequencing Assay Market
Includes market sizing, segmentation methodology, and regional forecast tables.
Regional Market Breakdown
*Sources: Market Research Future *
Competitive Landscape
*Sources: Market Research Future *
Outlook Through 2035
The convergence of NGS cost reduction, liquid biopsy acceptance, and AI-powered data analysis will define the miRNA sequencing assay market through 2035. Key trends shaping the market include:
Integration with Multi-Omics Approaches: miRNA sequencing increasingly integrated with genomics, proteomics, and metabolomics for comprehensive disease profiling, enabling systems biology understanding of complex diseases.
AI-Powered Data Analysis: Machine learning algorithms identifying novel miRNA biomarkers from large datasets, reducing analysis time and improving prediction accuracy for disease diagnosis and prognosis.
Point-of-Care Applications: Development of rapid, low-cost miRNA detection platforms for decentralized testing, enabling early cancer screening in community healthcare settings.
Extracellular Vesicle (EV) miRNA Analysis: Growing focus on exosome-derived miRNAs as stable, non-invasive biomarkers for liquid biopsy applications, particularly in oncology and neurology.
Regulatory Approvals for Clinical Use: Increasing number of miRNA-based diagnostic assays receiving FDA and CE-IVD clearance, validating clinical utility and expanding market access.
Emerging Applications in Drug Development: Pharmaceutical companies incorporating miRNA biomarkers into clinical trials for patient stratification, target engagement monitoring, and pharmacodynamic response assessment.
Vendors investing in NGS innovation, AI-powered analytics, point-of-care platforms, and regulatory-approved clinical assays will capture the highest-margin contracts as miRNA sequencing evolves from research tool to essential component of precision medicine diagnostics.
Access complete forecasts, segment analysis & competitive intelligence:
→ Purchase the Full MiRNA Sequencing Assay Market Report (2025-2035)
*10-year forecasts | Segment & application analysis | Regional data | Competitive landscape*
Keywords: MiRNA Sequencing | Assay Market | Liquid Biopsy | NGS | miRNA Biomarker | Cancer Diagnostics | Precision Medicine | Small RNA
© 2026 WiseGuy Reports (WGR) · All Rights Reserved · wiseguyreports.com
All market projections are forward-looking estimates sourced from WGR’s proprietary research reports and subject to revision.